156 Eukaryotic Epigenetic Gene Regulation
Learning Objectives
By the end of this section, you will be able to do the following:
- Explain how chromatin remodeling controls transcriptional access
- Describe how access to DNA is controlled by histone modification
- Describe how DNA methylation is related to epigenetic gene changes
Eukaryotic gene expression is more complex than prokaryotic gene expression because the processes of transcription and translation are physically separated. Unlike prokaryotic cells, eukaryotic cells can regulate gene expression at many different levels. Epigenetic changes are inheritable changes in gene expression that do not result from changes in the DNA sequence. Eukaryotic gene expression begins with control of access to the DNA. Transcriptional access to the DNA can be controlled in two general ways: chromatin remodeling and DNA methylation. Chromatin remodeling changes the way that DNA is associated with chromosomal histones. DNA methylation is associated with developmental changes and gene silencing.
Epigenetic Control: Regulating Access to Genes within the Chromosome
The human genome encodes over 20,000 genes, with hundreds to thousands of genes on each of the 23 human chromosomes. The DNA in the nucleus is precisely wound, folded, and compacted into chromosomes so that it will fit into the nucleus. It is also organized so that specific segments can be accessed as needed by a specific cell type.
The first level of organization, or packing, is the winding of DNA strands around histone proteins. Histones package and order DNA into structural units called nucleosome complexes, which can control the access of proteins to the DNA regions (Figure 16.7a). Under the electron microscope, this winding of DNA around histone proteins to form nucleosomes looks like small beads on a string (Figure 16.7b).
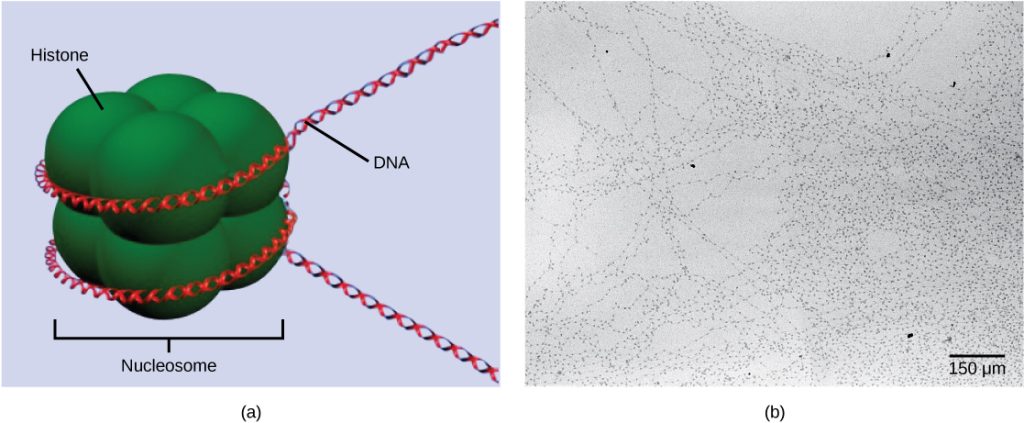
These beads (histone proteins) can move along the string (DNA) to expose different sections of the molecule. If DNA encoding a specific gene is to be transcribed into RNA, the nucleosomes surrounding that region of DNA can slide down the DNA to open that specific chromosomal region and allow for the transcriptional machinery (RNA polymerase) to initiate transcription (Figure 16.8).
Visual Connection
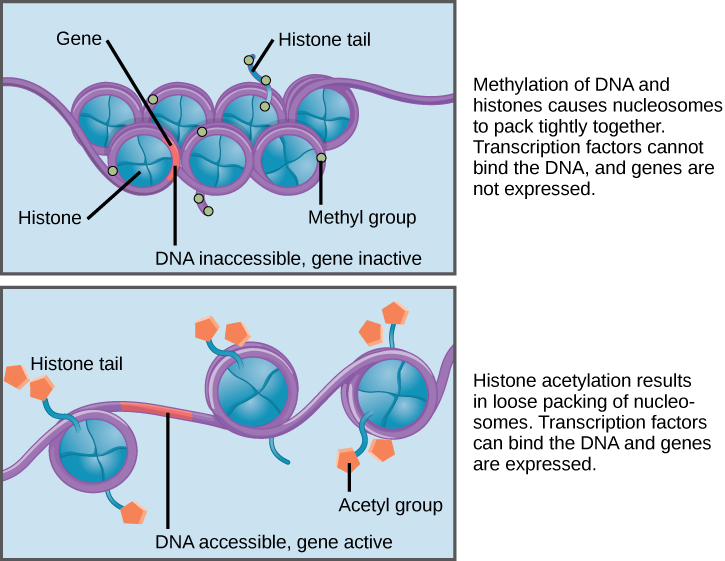
How closely the histone proteins associate with the DNA is regulated by signals found on both the histone proteins and on the DNA. These signals are functional groups added to histone proteins or to DNA and determine whether a chromosomal region should be open or closed (Figure 16.9 depicts modifications to histone proteins and DNA). These tags are not permanent, but may be added or removed as needed. Some chemical groups (phosphate, methyl, or acetyl groups) are attached to specific amino acids in histone “tails” at the N-terminus of the protein. These groups do not alter the DNA base sequence, but they do alter how tightly wound the DNA is around the histone proteins. DNA is a negatively charged molecule and unmodified histones are positively charged; therefore, changes in the charge of the histone will change how tightly wound the DNA molecule will be. By adding chemical modifications like acetyl groups, the charge becomes less positive, and the binding of DNA to the histones is relaxed. Altering the location of nucleosomes and the tightness of histone binding opens some regions of chromatin to transcription and closes others.
The DNA molecule itself can also be modified by methylation. DNA methylation occurs within very specific regions called CpG islands. These are stretches with a high frequency of cytosine and guanine dinucleotide DNA pairs (CG) found in the promoter regions of genes. The cytosine member of the CG pair can be methylated (a methyl group is added). Methylated genes are usually silenced, although methylation may have other regulatory effects. In some cases, genes that are silenced during the development of the gametes of one parent are transmitted in their silenced condition to the offspring. Such genes are said to be imprinted. Parental diet or other environmental conditions may also affect the methylation patterns of genes, which in turn modifies gene expression. Changes in chromatin organization interact with DNA methylation. DNA methyltransferases appear to be attracted to chromatin regions with specific histone modifications. Highly methylated (hypermethylated) DNA regions with deacetylated histones are tightly coiled and transcriptionally inactive.
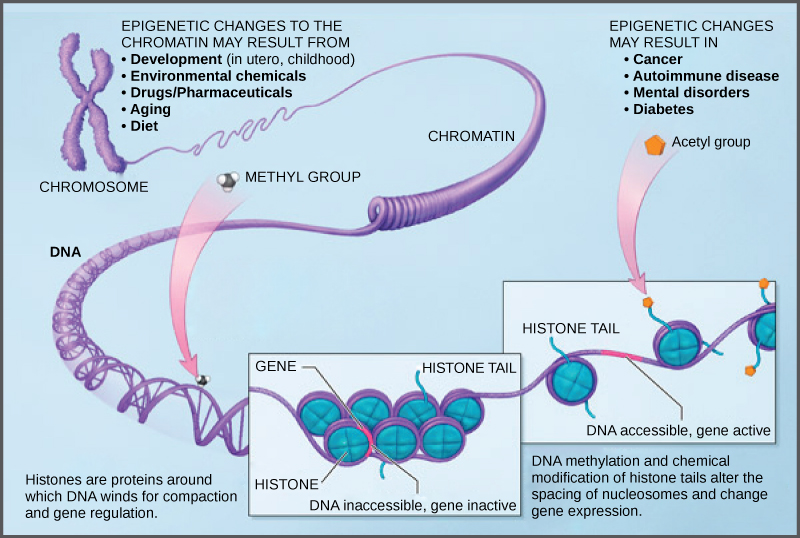
Epigenetic changes are not permanent, although they often persist through multiple rounds of cell division and may even cross generational lines. Chromatin remodeling alters the chromosomal structure (open or closed) as needed. If a gene is to be transcribed, the histone proteins and DNA in the chromosomal region encoding that gene are modified in a way that opens the promoter region to allow RNA polymerase and other proteins, called transcription factors, to bind and initiate transcription. If a gene is to remain turned off, or silenced, the histone proteins and DNA have different modifications that signal a closed chromosomal configuration. In this closed configuration, the RNA polymerase and transcription factors do not have access to the DNA and transcription cannot occur (Figure 16.9).
Link to Learning
View this video that describes how epigenetic regulation controls gene expression.

